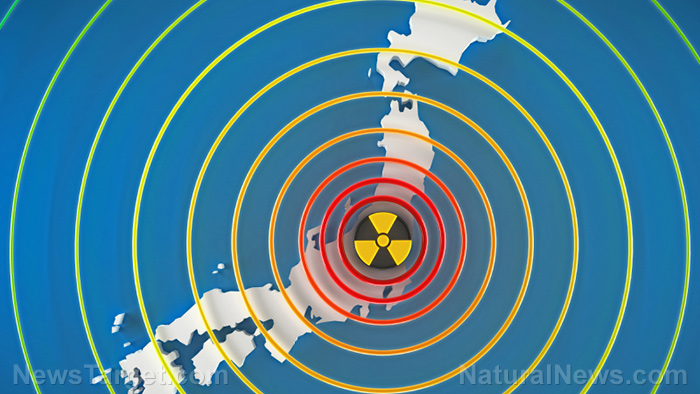
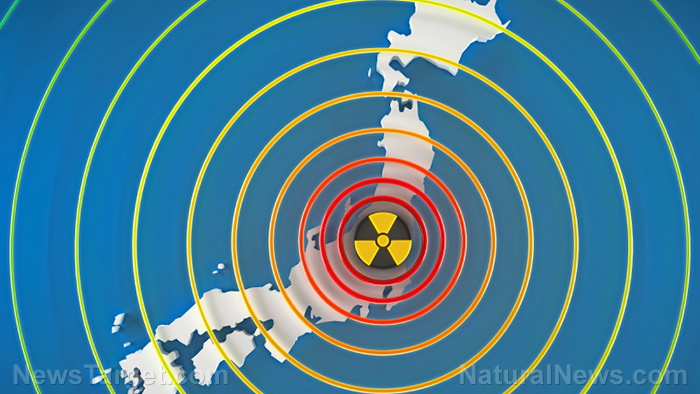
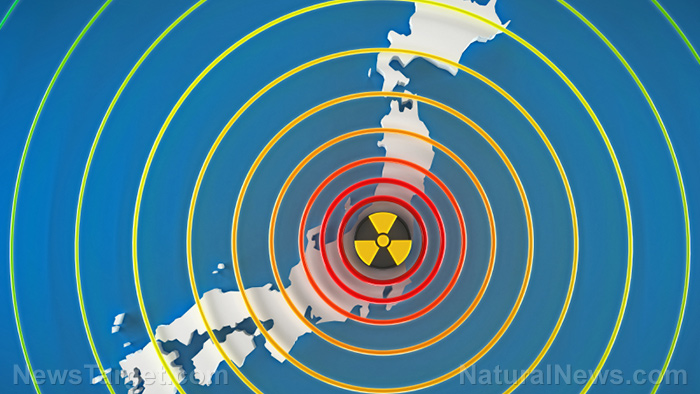
Fukushima fallout has condensed into glass microparticles…now too concentrated to dissolve easily
07/14/2016 / By Vicki Batts

Shocking new research has revealed that most of the nuclear fallout from Fukushima concentrated and deposited itself into microscopic, non-soluble glass particles in downtown Tokyo just days after the accident occurred. This glassy “soot” simply could not be dissolved by rain or running water — meaning it stayed in the environment unless it was physically removed.
These little particles also contained concentrated, radioactive cesium isotopes. This also means that, in many cases, dose effects of the fallout are still unclear. These results were announced at the Goldschmidt Geochemistry Conference in Yokohama, Japan in mid-June 2016. Five years after the accident, and we are still discovering how damaging its effects have been to the environment.
On March 11, 2011, massive flooding at the Fukushima-Daiichi Nuclear Power Plant following a catastrophic earthquake resulted in the devastating release of radioactive materials in large quantities. Naturally, this radioactive material continues to have a persistent presence in the environment. The radioactive isotope cesium-137, for example, has a half-life of 30 years.
Japanese geo-chemists from Kyushu University collected samples from an area within 230 kilometers from the power plant. Because cesium is water-soluble, the scientists expected that most of the cesium would have dissipated by now, hopefully flushed away by rainwater. Unfortunately, they discovered that most cesium from the fallout actually fell to the ground as microparticles encased in glassy, protective coverings that were formed when the reactor melted down.
As if that isn’t disturbing enough, as the cesium was being encased in a silicone oxide glass, it was actually concentrated, making it even more potent. Because of each particle’s high cesium content, the radioactivity per unit is between 10 million and 100 million times higher than the average cesium radioactivity found in the soil surrounding Fukushima.
In addition to worries about cesium contamination, in April of 2016, Japan also revealed plans to release 1,000 tanks-worth of tritium-contaminated water into the ocean. Tritium is another type of radioactive material, one that is near impossible to remove. Three hundred tonnes of contaminated water are produced daily, and they are running out of room to store it. So, why not just flush it all into the ocean, right?
Many officials have voiced concerns over this confounding plan, including the U.S. EPA, but some believe it’s totally safe. For example, the chairman of the Nuclear Regulation Authority, Shunichi Tanaka, told reporters, “Tritium is so weak in its radioactivity it won’t penetrate plastic wrapping.”
It is true that tritium is not capable of penetrating beyond the first layer of human skin — if it is merely present in the environment. However, most tritium exposure comes from contaminated water and food, and that, my friend, is where the trouble begins. The U.S. Nuclear Regulatory Commission states that once a person has consumed tritium, it is quickly dispersed into soft tissues. After ten days, about half of the tritium will be excreted. It is worth noting that tritium is a radioactive form of hydrogen, and it too can bond with oxygen to form water. Tritiated water is radioactive but chemically identical to normal drinking water, and the tritium cannot be filtered out.
So, the radioactive glass microparticles can’t be broken down, and the tritium can’t be filtered out of the water. Sure sounds like trouble, doesn’t it?
Sources:
Submit a correction >>
Tagged Under:
Fukushima, radiation, toxic soil, toxic water
This article may contain statements that reflect the opinion of the author
RECENT NEWS & ARTICLES
COPYRIGHT © 2017 FUKUSHIMAWATCH.COM
All content posted on this site is protected under Free Speech. FukushimaWatch.com is not responsible for content written by contributing authors. The information on this site is provided for educational and entertainment purposes only. It is not intended as a substitute for professional advice of any kind. FukushimaWatch.com assumes no responsibility for the use or misuse of this material. All trademarks, registered trademarks and service marks mentioned on this site are the property of their respective owners.